From Wikipedia, the free encyclopedia
In chemistry, an alcohol is any organic compound in which a hydroxyl group (–OH) is bound to a carbon atom of an alkyl or substituted alkyl group. An important group of acohols is formed by the simple acyclic alcohols, the general formula for which is CnH2n+1OH. Of those, ethanol (C2H5OH) is the type of alcohol found in
Other alcohols are usually described with a clarifying adjective, as in isopropyl alcohol (propan-2-ol) or wood alcohol (methyl alcohol, or methanol). The suffix -ol appears in the IUPAC chemical name of all alcohols.
There are three major subsets of alcohols: primary (1°), secondary (2°) and tertiary (3°), based upon the number of carbon atoms the C-OH group’s carbon (shown in red) is bonded to. Ethanol is a simple ‘primary’ alcohol. The simplest secondary alcohol is isopropyl alcohol (propan-2-ol), and a simple tertiary alcohol is tert-butyl alcohol (2-methylpropan-2-ol).
Contents[hide] |
Simple alcohols
The simplest and most commonly used alcohols are methanol and ethanol. Methanol was formerly obtained by the distillation of wood and called “wood alcohol.”
One sip of methanol (as little as 10ml) can cause permanent blindness by destruction of the optic nerve.[1]
Apart from its familiar role in
Two other alcohols whose uses are relatively widespread (though not so much as those of methanol and ethanol) are propanol and butanol. Like ethanol, they can be produced by fermentation processes. (However, the fermenting agent is a bacterium, Clostridium acetobutylicum, that feeds on cellulose, not sugars like the Saccharomyces yeast that produces ethanol.)
Nomenclature
Systematic names
In the
Some examples of simple alcohols and how to name them:
Common names for alcohols usually takes name of the corresponding alkyl group and add the word “alcohol”, e.g. methyl alcohol, ethyl alcohol or tert-butyl alcohol. Propyl alcohol may be n-propyl alcohol or isopropyl alcohol depending on whether the hydroxyl group is bonded to the 1st or 2nd carbon on the propane chain. Isopropyl alcohol is also occasionally called sec-propyl alcohol.
As mentioned above alcohols are classified as primary (1°), secondary (2°) or tertiary (3°), and common names often indicate this in the alkyl group prefix. For example (CH3)3COH is a tertiary alcohol is commonly known as tert-butyl alcohol. This would be named 2-methylpropan-2-ol under IUPAC rules, indicating a propane chain with methyl and hydroxyl groups both attached to the middle (#2) carbon.
Primary alcohol (1°)– Have general formulas RCH2OH Secondary alcohol (2°)– Have general formulas RR’CHOH Tertiary alcohol (3°)– Have general formulas RR’RCOH Hydrogen bond strength order: 1°>2°>3° Boiling point order: 1°>2°>3° Acidity order: 1°>2°>3°
Etymology
The word alcohol appears in English in the 16th century, loaned via French from medical Latin, ultimately from the
ال al is Arabic for the definitive article, the in English.
The current Arabic name for alcohol is الكحول al-kuḥūl, re-introduced from western usage.
kuḥl was the name given to the very fine powder, produced by the sublimation of the natural mineral stibnite to form antimony sulfide Sb2S3 (hence the essence or “spirit” of the substance), which was used as an antiseptic and eyeliner.
Bartholomew Traheron in his 1543 translation of John of Vigo introduces the word as a term used by “barbarous” (
- the barbarous auctours use alcohol, or (as I fynde it sometymes wryten) alcofoll, for moost fine poudre.
William Johnson in his 1657 Lexicon Chymicum glosses the word as antimonium sive stibium. By extension, the word came to refer to any fluid obtained by distillation, including “alcohol of wine”, the distilled essence of wine.
Physical and chemical properties
Alcohols have an odor that is often described as “biting” and as “hanging” in the nasal passages.
The
Because of
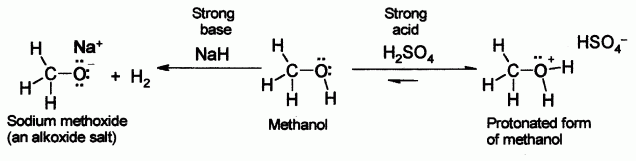
Alcohols, like water, can show either acidic or basic properties at the O-H group. With a
Meanwhile the oxygen atom has lone pairs of nonbonded electrons that render it weakly basic in the presence of strong acids such as sulfuric acid. For example, with methanol:
Alcohols can also undergo
Applications
Total recorded
Alcohols can be used as a beverage (ethanol only), as fuel and for many scientific, medical, and industrial utilities. Ethanol in the form of alcoholic beverages has been consumed by humans since pre-historic times. A 50% v/v solution of ethylene glycol in water is commonly used as an antifreeze.
Some alcohols, mainly ethanol and methanol, can be used as an alcohol fuel. Fuel performance can be increased in forced induction internal combustion engines by injecting alcohol into the air intake after the turbocharger or supercharger has pressurized the air. This cools the pressurized air, providing a denser air charge, which allows for more fuel, and therefore more power.
Alcohols have applications in industry and science as reagents or solvents. Because of its low toxicity and ability to dissolve
Ethanol can be used as an antiseptic to disinfect the skin before injections are given, often along with iodine. Ethanol-based soaps are becoming common in restaurants and are convenient because they do not require drying due to the volatility of the compound. Alcohol is also used as a preservative for specimens.
Alcohol gels have become common as hand sanitizers.
Production
Industrially alcohols are produced in several ways:
- By fermentation using glucose produced from sugar from the hydrolysis of starch, in the presence of yeast and temperature of less than 37°C to produce ethanol. For instance the conversion of invertase to glucose and fructose or the conversion of glucose to zymase and ethanol.
- By direct hydration using ethylene (ethylene hydration[4]) or other alkenes from cracking of fractions of distilled
crude oil.
Endogenous
Several of the benign bacteria in the intestine use fermentation as a form of anaerobic respiration. This metabolic reaction produces ethanol as a waste product, just like
Laboratory synthesis
Several methods exist for the preparation of alcohols in the laboratory.
- Primary
alkyl halides react with aqueous NaOH or KOH mainly to primary alcohols in nucleophilic aliphatic substitution. (Secondary and especially tertiary alkyl halides will give the elimination (alkene) product instead). Aldehydes or ketones are reduced with sodium borohydride or lithium aluminium hydride (after an acidic workup). Another reduction by aluminiumisopropylates is the Meerwein-Ponndorf-Verley reduction. Alkenes engage in an acid catalysed hydration reaction using concentrated sulfuric acid as a catalyst which gives usually secondary or tertiary alcohols. The hydroboration-oxidation and oxymercuration-reduction of alkenes are more reliable in organic synthesis. Alkenes react with NBS and water in halohydrin formation reaction Grignard reagents react with carbonyl groups to secondary and tertiary alcohols. Related reactions are the Barbier reaction and the Nozaki-Hiyama reaction. - Noyori asymmetric hydrogenation is the asymmetric reduction of β-keto-esters
- Amines can be converted to
diazonium salts which are then hydrolyzed.
The formation of a secondary alcohol via reduction and hydration is shown:
Reactions
Deprotonation
Alcohols can behave as weak acids, undergoing deprotonation. The deprotonation reaction to produce an alkoxide salt is either performed with a strong base such as sodium hydride or n-butyllithium, or with sodium or potassium metal.
- 2 R-OH + 2Na → 2R-O−Na + H2
- E.g. 2 CH3CH2-OH + 2 Na → 2 CH3-CH2-O−Na + H2
Water is similar in
- R-OH + NaOH <=> R-O–Na+ + H2O (equilibrium to the left)
It should be noted, though, that the bases used to deprotonate alcohols are strong themselves. The bases used and the alkoxides created are both highly moisture sensitive chemical reagents.
The acidity of alcohols is also affected by the overall stability of the alkoxide ion.
With
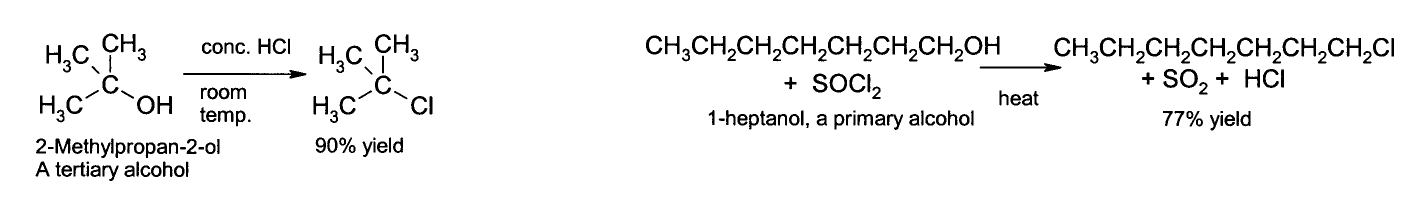
Nucleophilic substitution
The OH group is not a good leaving group in nucleophilic substitution reactions, so neutral alcohols do not react in such reactions. However, if the oxygen is first protonated to give R−OH2+, the leaving group (
Alcohols may likewise be converted to alkyl bromides using hydrobromic acid or phosphorus tribromide, for example:
- 3 R-OH + PBr3 → 3 RBr + H3PO3
In the
Dehydration
Alcohols are themselves nucleophilic, so R−OH2+ can react with ROH to produce ethers and water in a dehydration reaction, although this reaction is rarely used except in the manufacture of diethyl ether.
More useful is the E1 elimination reaction of alcohols to produce alkenes. The reaction generally obeys
This is a diagram of acid catalysed dehydration of ethanol to produce
A more controlled elimination reaction is the Chugaev elimination with carbon disulfide and iodomethane.
Esterification
To form an ester from an alcohol and a carboxylic acid the reaction, known as
- R-OH + R’-COOH → R’-COOR + H2O
In order to drive the equilibrium to the right and produce a good yield of ester, water is usually removed, either by an excess of H2SO4 or by using a Dean-Stark apparatus. Esters may also be prepared by reaction of the alcohol with an
Other types of ester are prepared similarly- for example tosyl (tosylate) esters are made by reaction of the alcohol with p-
Oxidation
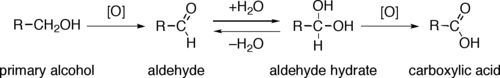
Primary alcohols (R-CH2-OH) can be oxidized either to aldehydes (R-CHO) or to carboxylic acids (R-CO2H), while the oxidation of secondary alcohols (R1R2CH-OH) normally terminates at the ketone (R1R2C=O) stage. Tertiary alcohols (R1R2R3C-OH) are resistant to oxidation.
The direct oxidation of primary alcohols to carboxylic acids normally proceeds via the corresponding aldehyde, which is transformed via an aldehyde hydrate (R-CH(OH)2) by reaction with water before it can be further oxidized to the carboxylic acid.
Often it is possible to interrupt the oxidation of a primary alcohol at the aldehyde level by performing the reaction in absence of water, so that no aldehyde hydrate can be formed.
Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the oxidation of secondary alcohols to ketones. These include:
- Chromium-based reagents, such as Collins reagent (CrO3·Py2),
PDC or PCC. - Activated DMSO, resulting from reaction of DMSO with electrophiles, such as oxalyl chloride (Swern oxidation), a carbodiimide (
Pfitzner-Moffatt oxidation) or the complex SO3·Py (Parikh-Doering oxidation). - Hypervalent iodine compounds, such as
Dess-Martin periodinane or 2-Iodoxybenzoic acid. - Catalytic TPAP in presence of excess of NMO (
Ley oxidation). - Catalytic TEMPO in presence of excess bleach (
NaOCl) (Anelli’s oxidation).
Allylic and benzylic alcohols can be oxidized in presence of other alcohols using certain selective oxidants such as manganese dioxide (MnO2).
Reagents useful for the oxidation of secondary alcohols to ketones, but normally inefficient for oxidation of primary alcohols to aldehydes, include chromium trioxide (CrO3) in a mixture of sulfuric acid and acetone (Jones oxidation) and certain ketones, such as cyclohexanone, in the presence of aluminium isopropoxide (Oppenauer oxidation).
The direct oxidation of primary alcohols to carboxylic acids can be carried out using:
- Potassium permanganate (KMnO4).
- Jones oxidation.
PDC in DMF. - Heyns oxidation.
- Ruthenium tetroxide (RuO4).
- TEMPO.
Alcohols possessing two hydroxy groups located on adjacent carbons —that is, 1,2-diols— suffer oxidative breakage at a carbon-carbon bond with some oxidants such as sodium periodate (NaIO4) or
Toxicity

Most significant of the possible
Ethanol in
The
An effective treatment to prevent formaldehyde toxicity after methanol ingestion is to administer ethanol. Alcohol dehydrogenase has a higher affinity for ethanol, thus preventing methanol from binding and acting as a
Methanol itself, while poisonous, has a much weaker sedative effect than ethanol. Some longer-chain alcohols such as
Occurrence in nature
Alcohol has been found outside the Solar system. It can be found in low densities in star and planetary system forming regions of space.
See
- http://www.springerlink.com/content/u3p6612j611w2hqv/
- http://www.physics.uq.edu.au/people/ross/phys2080/ism/ism.htm (species found)
- Astrophysical maser (species found)
See also
- Alcohol fuel
- Alcoholic beverage
- Blood alcohol content
- Breathalyzer
- Fatty alcohol
- Fetal alcohol syndrome
- History of alcohol
- List of countries by alcohol consumption
- Oxidation of primary alcohols to carboxylic acids
- Oxidation of secondary alcohols to ketones
- Phenols
- Rubbing alcohol
- Sugar alcohol
- Surrogate alcohol
- Transesterification
- Cooking with alcohol
References
- ^ a b “Methanol and Blindness“. Ask A Scientist, Chemistry Archive. http://www.newton.dep.anl.gov/askasci/chem03/chem03561.htm
. Retrieved 22 May 2007. - ^ a b William Reusch. “Alcohols“. VirtualText of Organic Chemistry. http://www.cem.msu.edu/~reusch/VirtualText/alcohol1.htm#alcnom
. Retrieved 2007-09-14. - ^ Global Status Report on Alcohol 2004
- ^ Lodgsdon, J.E. (1994). “Ethanol.” In J.I. Kroschwitz (Ed.) Encyclopedia of Chemical Technology, 4th ed. vol. 9, p. 820. New York: John Wiley & Sons.
- ^ Robert S. Gable (2004). “Comparison of acute lethal toxicity of commonly abused psychoactive substances” (reprint). Addiction 99 (6): 686–696. doi:10.1111/j.1360-0443.2004.00744.x. http://web.cgu.edu/faculty/gabler/toxicity%20Addiction%20offprint.pdf.
- ^ Zimmerman HE, Burkhart KK, Donovan JW. Ethylene glycol and methanol poisoning: diagnosis and treatment. Journal of Emergency Nursing. 1999 Apr;25(2):116-20. PMID 10097201
- ^ Lobert S. Ethanol, isopropanol, methanol, and ethylene glycol poisoning. Critical Care Nurse. 2000 December;20(6):41-7. PMID 11878258
- ^ McKee M, Suzcs S, Sárváry A, Adany R, Kiryanov N, Saburova L, Tomkins S, Andreev E, Leon DA. The composition of surrogate alcohols consumed in Russia. Alcoholism, Clinical and Experimental Research. 2005 October;29(10):1884-8. PMID 16269919
- ^ Bunc M, Pezdir T, Mozina H, Mozina M, Brvar M. Butanol ingestion in an airport hangar. Human and Experimental Toxicology. 2006 Apr;25(4):195-7. PMID 16696295
- ^ Woo KL. Determination of low molecular weight alcohols including fusel oil in various samples by diethyl ether extraction and capillary gas chromatography. Journal of AOAC International. 2005 September-October;88(5):1419-27. PMID 16385992
- ^ Lachenmeier DW, Haupt S, Schulz K. Defining maximum levels of higher alcohols in alcoholic beverages and surrogate alcohol products. Regulatory Toxicology and Pharmacology. 2008 Apr;50(3):313-21. PMID 18295386
- ^ Hori H, Fujii W, Hatanaka Y, Suwa Y. Effects of fusel oil on animal hangover models. Alcohol Clinical and Experimental Research. 2003 Aug;27(8 Suppl):37S-41S. PMID 12960505
- ^ Wiernikowski A, Piekoszewski W, Krzyzanowska-Kierepka E, Gomułka E. Acute oral poisoning with isopropyl alcohol in alcoholics. (Polish) Przeglad Lekarski. 1997;54(6):459-63. PMID 9333902
- ^ Mańkowski W, Klimaszyk D, Krupiński B. How to differentiate acute isopropanol poisoning from ethanol intoxication? — a case report. (Polish) Przeglad Lekarski 2000;57(10):588-90. PMID 11199895
- ^ Bogomolova IN, Bukeshov MK, Bogomolov DV. The forensic medical diagnosis of intoxication of alcohol surrogates by morphological findings. (Russian) Sudebno Meditsinskaia Ekspertiza. 2004 September-October;47(5):22-5. PMID 15523882
Bibliography
- Metcalf, Allan A. (1999). The World in So Many Words. Houghton Mifflin. ISBN 0395959209.